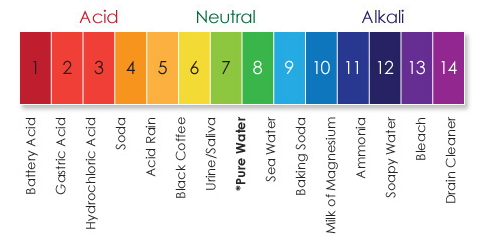
Phenolphthalein is a pH indicator, but it only turns colors in reaction to bases. Refill the glasses one last time-the liquid will be colorless again! What Happened: Now pour all five glasses back in the pitcher. Refill the remaining four glasses – the water will be red!ĥ. Fill all the glasses with water from the pitcher, then pour all of them back in the pitcher except for the glass with vinegar.Ĥ. Add a few drops of water to the first glass and stir to dissolve the sodium carbonate.ģ. In the first glass put a little less than 1/8 teaspoon of sodium carbonate, in the second put 6 drops of phenolphthalein solution, and in the third put three droppers-full of vinegar.Ģ. 5 glasses and a non-see-through pitcher of waterġ.> Check out our project video to see this trick in action! Make ordinary water turn bright pink and then back to clear! This makes a great “magic trick” to impress your friends – just be careful no one mistakes it for fruit punch and drinks any! You can also make your own pH indicator-use a blender to mix one part chopped red cabbage with two parts boiling water and use the juice to test different solutions.Īcids will turn the pigments in the indicator to a reddish color bases will turn the pigments bluish or yellow-green. Human blood has an ideal pH of 7.4 even slight fluctuations can seriously affect our bodies. Here are the pH levels of some other substances that you might test: Use the litmus paper to determine which are acids and which are bases. Try testing window cleaner, toilet bowl cleaner, orange juice, and apple juice-pour a little of each into separate test tubes or small glasses or jars. If you have red and blue litmus paper, you can test different solutions for whether they are acids or bases.īlue litmus paper turns red when a solution is acidic red litmus paper turns blue in basic solutions. Litmus is a natural acid-base indicator extracted from a type of lichen. Do the following projects to explore the cool effects of pH. Neutral solutions (like distilled water) with a balanced number of H+ and OH- ions have a pH of 7. Acids have a pH below 7 bases have a pH above. The pH scale is used to measure how acidic or basic a solution is.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
